Features You'll Love
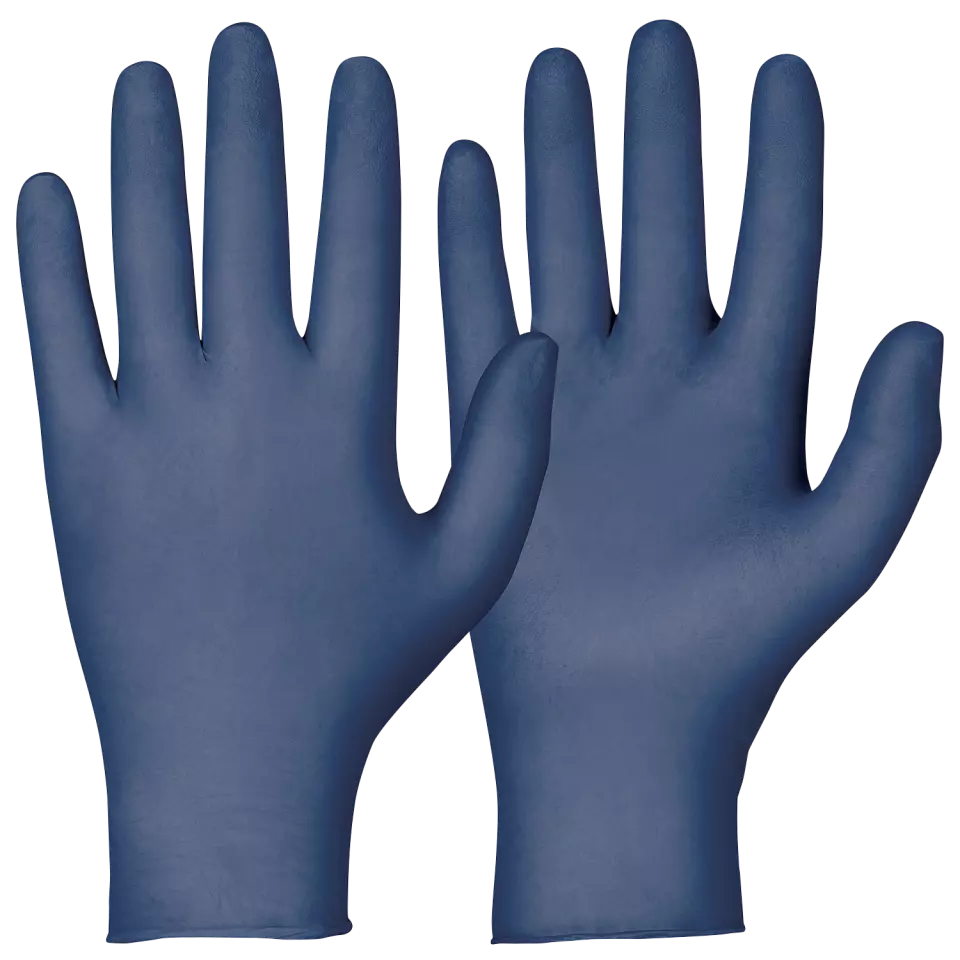
Fingertips · Textured
Describes the surface texture of the fingertip area, affecting grip strength and tactile sensitivity during use.
58,17 €
Price per 10 packages (1 000 pcs)
5,82 € / 100 pcs
Shipping fee is 7,95 € for orders under 750,00 €
Product description
These premium medical-grade nitrile gloves offer exceptional sensitivity and protection as the thinnest option in Granberg's Accelerators Free range. Engineered with micro-textured fingertips for superior grip in both wet and dry conditions, these gloves provide outstanding resistance to various substances including grease, fats, and detergents. The powder-free design and complete absence of chemical accelerators make them ideal for sensitive applications while maintaining excellent durability.
Product Features:
- 100% accelerator-free nitrile construction
- Micro-textured fingertips for optimal grip
- Powder-free design
- Tight-fitting for maximum precision
- Waterproof protection
Technical Details:
- Thickness: Min 0.05 mm (palm)
- Length: 25 cm
- Weight: 3.2 ± 0.5 g
- AQL Level: 1.5
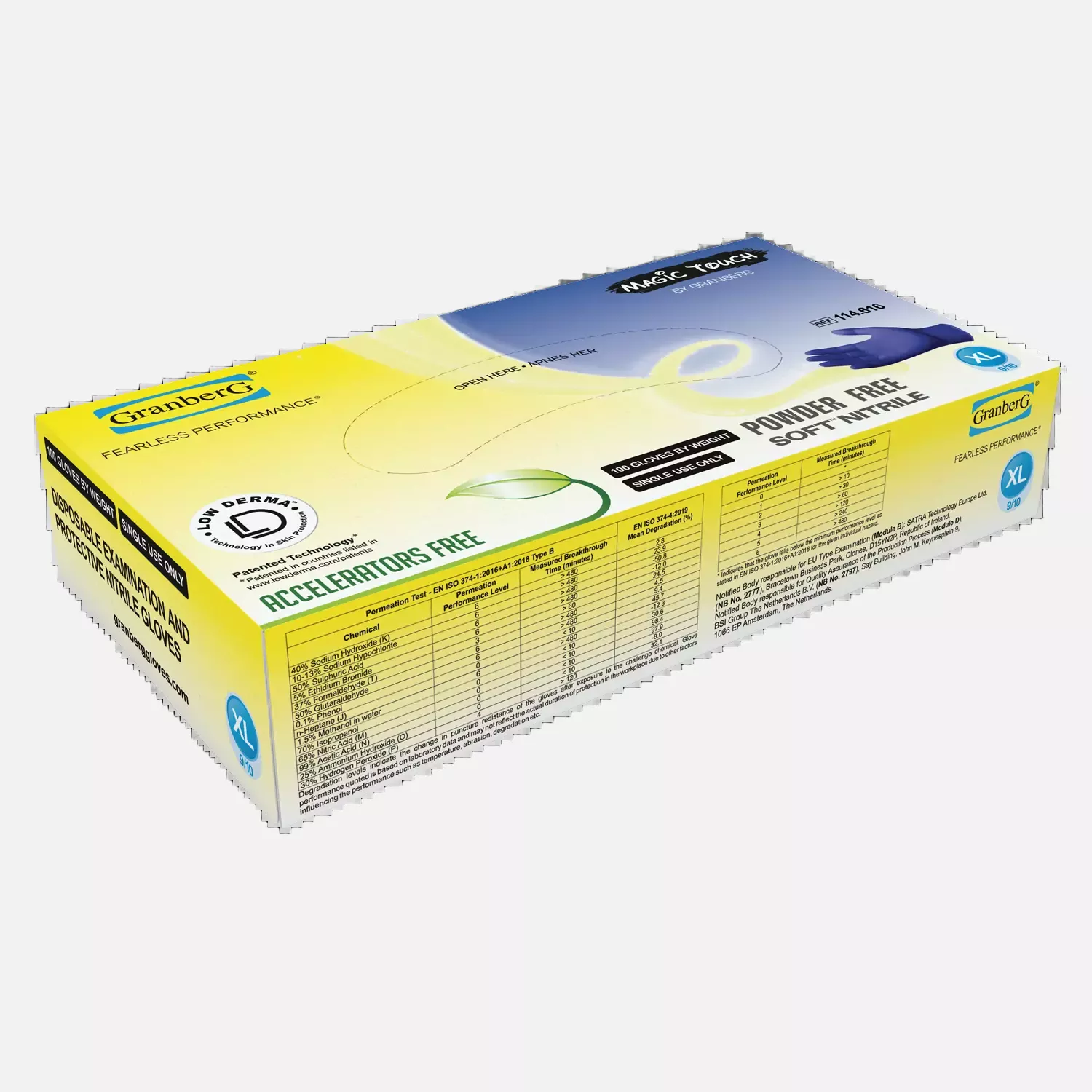
Chemical Resistance:
- 40% Sodium Hydroxide
- 30% Hydrogen Peroxide
- 37% Formaldehyde
Standards:
- CE Category III (Notified body 2797)
- EN 455 Medical use approved
- EN 1186 Food contact approved
- EN ISO 21420:2020
- ASTM D6978-05 Chemotherapy drug permeation
- MDR compliant
Recommended Applications:
- Medical procedures
- Direct contact with all types of foodstuffs
- Laboratory work
- Chemical handling