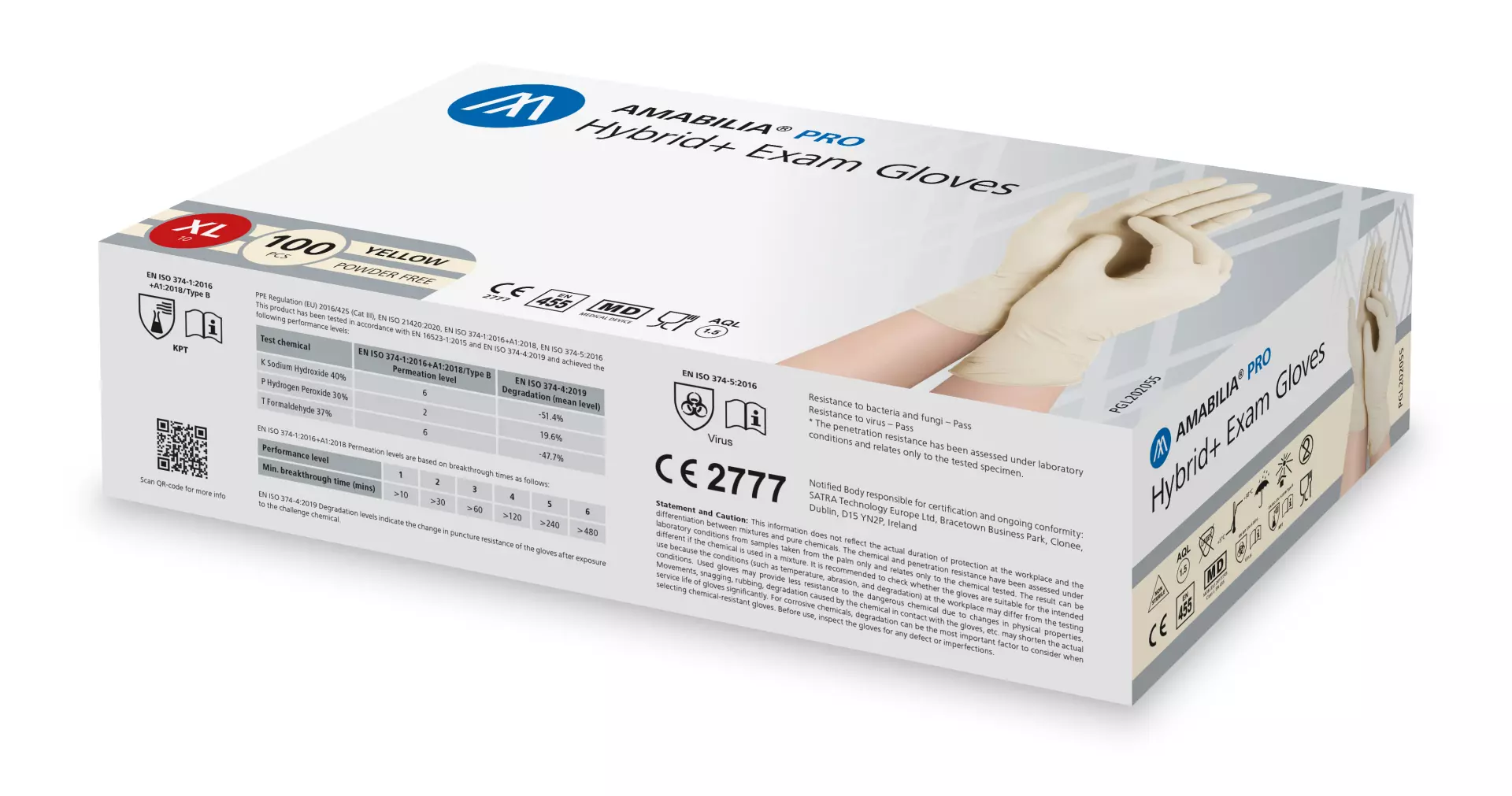
Amabilia Pro Hybrid+ Exam Glove Yellow
Amabilia Pro Hybrid+ Exam Glove Yellow
5 / 5





Features You'll Love
Cuff Style · Rolled
The design of the wrist opening, affecting ease of putting gloves on, comfort, and how securely they stay in place during use.
The design of the wrist opening, affecting ease of putting gloves on, comfort, and how securely they stay in place during use.
The design of the wrist opening, affecting ease of putting gloves on, comfort, and how securely they stay in place during use.
53,05 €
Price per 10 packages (1 000 pcs)
5,31 € / 100 pcs
Shipping fee is 7,95 € for orders under 600,00 €
Product description
The product description has not been specified
The product description has not been specified
The product description has not been specified
The texture of the glove exterior, affecting grip strength and handling capability for different tasks and working conditions.
The design of the wrist opening, affecting ease of putting gloves on, comfort, and how securely they stay in place during use.
Total mass of the glove package, useful for shipping calculations, storage planning, and comparing product quantities across different brands.
Indicates how long gloves maintain their protective properties and quality when stored properly, typically measured in years from manufacture date.
Indicates the maximum percentage of defective gloves acceptable in quality testing, with lower numbers representing higher quality standards.
Measures the material thickness at the palm area, affecting protection level and tactile sensitivity during use.
Measures how far the glove extends up the wrist and forearm, determining the level of coverage and protection provided during use.
Measures the material thickness at the fingertips, affecting tactile sensitivity, dexterity, and protection level during use.
Measures the thickness of the glove's wrist/forearm extension, affecting durability, tear resistance, and protection level during use.
Describes the surface texture of the fingertip area, affecting grip strength and tactile sensitivity during use.
The visual appearance of the glove material, ranging from basic colors to specialized options for different professional and aesthetic preferences.
Protects sensitive surfaces from silicone contamination, ensuring product integrity and quality.
Provides comfort and peace of mind, free from natural rubber latex to protect sensitive skin.
Complete finger coverage ensures full protection and hygiene for your hands, maintaining dexterity for all tasks.
Your go-to gloves for diverse tasks, offering reliable protection and convenience across many applications.
Indicates whether gloves contain powder on the interior surface to aid donning, affecting ease of use and contamination control requirements.
- Chemical Resistance
- Clean Room
- Food Service
- Medical Protection
- Antimicrobial Protection
- Hand Protection
Protective gloves with the EN ISO 21420:2020 rating are tested for general requirements, including design, construction, the absence of harmful chemicals, correct sizing, and dexterity. This ensures the gloves are safe and comfortable to wear, fit correctly, and won't cause adverse skin reactions.
Test results
Ensures gloves are designed for comfort, fit, and dexterity without using harmful materials. It covers general requirements like safe pH levels and non-toxic dyes, forming the foundation for all protective gloves.
Gloves with the EN ISO 374-1:2016 rating are tested for resistance against dangerous chemicals and microorganisms, including penetration, permeation, and degradation. This means you can choose gloves designed to protect your hands from specific chemical hazards, ensuring safer use in various tasks.
Gloves with the EN 374-5:2016 rating are tested for resistance to penetration by bacteria and fungi, and potentially viruses. This means the gloves provide a protective barrier against microorganisms, helping to keep your hands safe from harmful biological agents.
Test results
This glove is tested to protect you from bacteria, fungi, and viruses. It provides a reliable barrier, having passed specific tests to ensure no leakage when exposed to these micro-organisms, making it suitable for handling contaminated materials.
This product protects you from bacteria and fungi. It passed leakage tests, making it a reliable barrier when handling materials where these micro-organisms are a risk, such as in cleaning or laboratory work.
Protective gloves with the EN ISO 374-2:2019 rating are tested for their resistance to penetration by dangerous chemicals and micro-organisms. This standard checks for leaks, holes, or imperfections, ensuring the gloves form an effective barrier to keep hazardous substances from reaching your skin.
Protective clothing, gloves, and footwear materials with the EN 16523-1:2015 rating are tested for resistance to permeation by hazardous liquid chemicals, specifically how long it takes for a chemical to pass through the material. This ensures the product provides a dependable barrier, offering a measurable level of safety when handling dangerous liquids.
Test results
This product’s material resists liquid chemical breakthrough. It is tested for how long it takes a chemical to move through the material on a molecular level, offering protection for a specified time during continuous contact.
Protective gloves with the EN 374-3:2003 rating are tested for their material's resistance to chemical permeation, meaning how long it takes for a hazardous chemical to pass through. This helps you choose gloves that offer a specific level of protection against chemicals, keeping your hands safe during use.
Medical gloves with the EN 455-3:2015 rating are tested for biological safety, including protein, endotoxin, chemical, and powder content. This ensures the gloves are safe for use, reducing risks of allergic reactions and irritation for consumers.
Single-use medical gloves with the EN 455-2:2015 rating are tested for physical properties like dimensions and strength. This ensures gloves fit correctly and are strong enough to provide reliable protection without tearing during use.
Test results
This product is tested for physical strength and durability to ensure reliable performance. It meets specific requirements for size, stretch, and force-at-break, providing adequate protection against cross-contamination for both the user and patient.
Medical gloves for single use with the EN 455-1:2020+A1:2022 rating are tested for freedom from holes and leakage. This ensures the gloves provide a reliable barrier, minimizing the risk of contamination for your safety and protection.
Test results
These gloves are tested for tiny pinholes to a specific quality level (AQL) to ensure a reliable barrier. A lower AQL number means a lower chance of defects, offering stronger protection against bacteria and viruses.
Single-use medical gloves with the EN 455-4:2009 rating are tested for their shelf life, ensuring they maintain properties like strength and barrier integrity over time. This means the gloves will remain effective and safe, providing reliable protection against infections until their expiration date when stored correctly.
Medical devices with the ISO 13485:2016 rating are tested for a quality management system that ensures consistent safety, quality, and regulatory compliance throughout their lifecycle. This provides assurance that the product is reliably produced to high international standards for patient use.
Test results
This product was made under a quality management system that ensures consistent design, production, and distribution. This focuses on the safety, performance, and regulatory compliance of medical devices throughout their entire lifecycle, from design to disposal.
Flexible tubes for products like cosmetics, food, and pharmaceuticals with the EN 12374:2009 rating are tested for consistent names and descriptions for the parts of these tubes. This helps ensure clear communication among manufacturers and suppliers, contributing to reliable and well-designed packaging for your everyday items.
Protective gloves with the EN 420:2003+A1:2009 rating are tested for general design, safe materials (innocuousness), water penetration, comfort, and dexterity. This ensures the gloves are safe, fit correctly, and allow you to perform tasks effectively without discomfort.
Laboratory chemicals and dyes with the EN ISO 19001:2013 rating are tested for the clear and consistent information provided by the manufacturer. This ensures that users, such as medical professionals, can achieve reliable and reproducible results when using these products in biological staining.
REACH stands for "Registration, Evaluation, Authorisation and Restriction of Chemicals" it is a regulation of the European Union that governs the registration, evaluation, authorization, and restriction of chemicals. The REACH regulation aims to ensure that the chemicals used in Europe are safe for human health and the environment. The regulation applies to all chemical substances, whether they are used in industrial processes or in everyday products such as cleaning products, paints, and cosmetics. Companies that manufacture, import or use chemicals in the EU must register the chemicals they produce or import, provide safety information and communicate the risks associated with the chemicals. This information will be used to assess the risks and determine if any further action is needed to protect human health and the environment.
Food safe refers to the safety of food products that are used or consumed by people. In Europe, food safety is regulated by the European Union (EU) and the European Food Safety Authority (EFSA). These organizations set standards and requirements for food products to ensure they are safe to eat. To be considered "food safe" in Europe, a product must meet these standards and be free of harmful substances. This includes being free of harmful bacteria, pesticides, and other contaminants. Food products that do not meet these standards cannot be sold or used in the EU.
MD stands for "Medical Device." It refers to any instruments, apparatus, machines, implants, or other similar or related articles that are intended to be used for human beings for the purpose of diagnosis, prevention, monitoring, treatment or alleviation of disease, injury or disability. In Europe, a MD Label is a special label that must be on all Medical Devices that are sold or used in the European Union (EU). The label must include information about the product, such as the name of the manufacturer, the intended use of the product and CE mark. To be able to sell or use a Medical Device in the EU, the device must meet certain standards and requirements set by the European Union and notified body.
PPE stands for "personal protective equipment." PPE Category 3 refers to equipment that is complex and provide the highest level of protection such as powered respirators, SCBA, and full body suits. In Europe, PPE Category 3 must meet certain safety standards set by the European Union, which means that it must be designed and manufactured to protect the user without causing harm. Companies that make or sell PPE must prove that it meets these standards. They also must have a quality management system in place, have to be audited regularly by a notified body and have to have a technical documentation.
Free delivery when you order more than 600,00 € from Amabilia Medical
Supplier shipping fee 7,95 €
Brand minimum 100,00 €
Other products you may like
Similar products you may like
Find +150,000 products from hundreds of brands